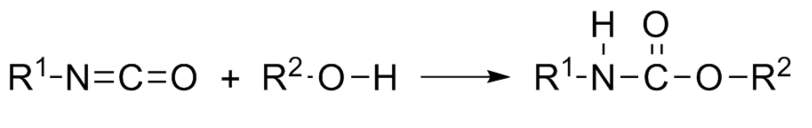General:
Like many plastics polymers, polyurethanes have emerged in all areas of our daily life through their nature, multiple properties, and their diversity of applications. Thermosetting their state, ie irreversible (unlike thermoplastics) does not prevent them from being used everywhere.
This versatility polyurethanes allows a wide range of applications, flexible foam for comfort and sound insulation, rigid foam thermal insulation, polyurethane compact … Changes in technology implementation and chemistry allows cycles short manufacturing, high reproducibility in large quantities. Their implementation has now reached a stage of maturity and consolidation.
All these qualities make it a major part of everyday life and portend that progress is still possible, both the materials side, that means the next transformation. With a growing market, polyurethane will be center of attention in research and development.
Detail:
Polyurethanes are polymers containing the grouping uréthane. We call urethane any compound produced by the reaction of an isocyanate and an alcohol according to the following reaction:
This bond results from the condensation reaction between a mobile hydrogen compound alcohol R-OH and R’-N = C = O isocyanate. To form a polyurethane, it is necessary reacting polyols (at least two alcohol functions per molecule) with polyisocyanates (at least two isocyanate functional groups per molecule).
The urethane function, obtained by the addition of an isocyanate with an alcohol, is known since the late nineteenth century and the reactions of isocyanates on various components were studied in the early twentieth century. These reactions are generally exothermic.
It is mainly the use of a wide range of types of polyols, molar masses and different features that could allow the development of the wide variety of polyurethanes.
Indeed, these polyols can be:
• long polyol polyesters resulting from polycondensation between a diacid and an excess of diol in the case of linear molecules or of the polyaddition of lactones by opening cycles as polycaprolactones,
• or long polyethers resulting from the polyaddition by opening ethers cycles (oxirane (s), oxetane, or tetrahydrofuran)
• other long diols and polyols such as polybutadienes, hydroxy, polycarbonates or of hydroxytelechelic polysiloxanes.
• very short diol called extenders or chain tensioners
• short polyols of functionalities greater than three called crosslinking or rétifiants.
The formulation is used to change the properties of polyurethanes through the use of many additives and fillers.
This impressive set of materials provides endless formulations.
It is this impressive diversity of raw materials that establishes(constitutes) the key of the diversity of polyurethanes both at the level of the structures and the applications.
So, polyurethanes constitute one of the vastest families of materials called “plastics”, although polyurethanes are more often structures thermodurcissables than thermoplastic:
• Flexible Foams of low density (10-80 kg / m3)
• flexible high density foam (> 100 kg / m3)
• Semi-rigid and micro cellular foams (400-800 kg / m3)
• rigid foam (30-80 kg / m3)
• high density rigid foam,
• Elastomers and thermoset casting masses,
• Thermoplastic Elastomers and plastomers,
• Textile fibers (types of elastane)
• Adhesives, paints, coatings and varnishes,
• Adhesives and binders.
The foams constitute a separate family, generating the highest production volumes, the rest of the polyurethanes is a commonly known CASE family by the industrial sector (Coatings, Adhesives, Sealants and Elastomers).